Overview: ChimerMarker - Automated Chimerism Analysis software
Product informationLicense Options Trial Request Technical Support

Automated Chimerism Analysis software
ChimerMarker® automated chimerism analysis software integrates speed and accuracy with a biologist-friendly interface. The software can be used to monitor chimerism level in both an allogeneic stem cells transplant (SCT) or a hematopoietic stem cells transplant (HSCT), bone marrow transplant (BMT, post bone marrow engraftment), and cord and peripheral blood stem cells transplant (PBSCT) samples.
Complete Chimerism Detection, Long-term Monitoring, and Quantification in one program!
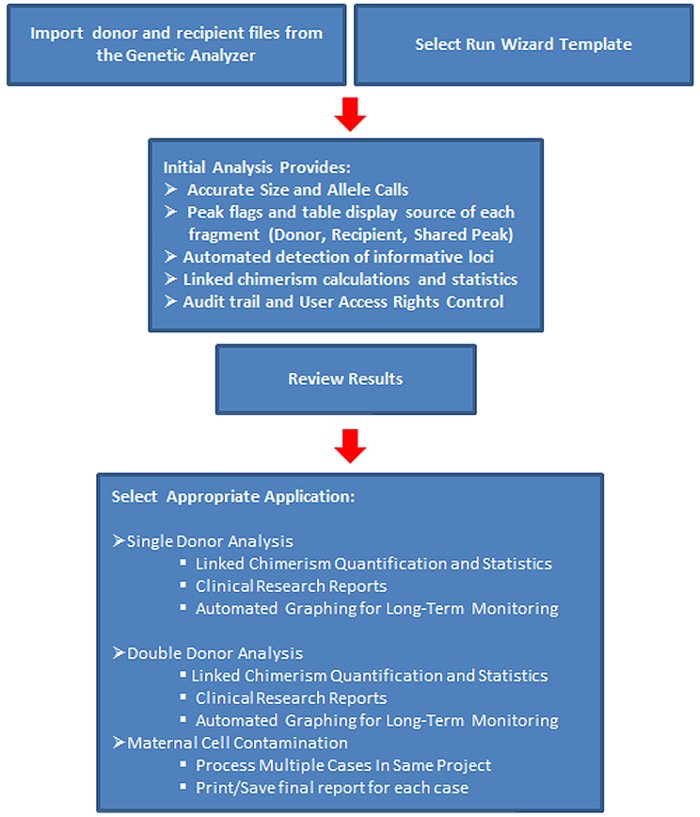
ChimerMarker software provides:
- Accurate, rapid genotyping and chimerism analysis with documented time savings of up to 85%
- Automatically identifies donor and recipient peaks in post-BMT samples
- Calculates percent chimerism and quality metrics for single donor or double donor cases
- Easily appends for longitudinal monitoring post-BMT
- Multi-lineage capabilities for chimerism analysis of T-cells, B-cells, and other cell type populations
ChimerMarker software includes functions for comparison of samples at different time points to conduct longitudinal studies for monitoring each individual and a comprehensive chimerism analysis report. The chimerism analysis performs repetitive calculations (using published methods. Dr. Don Kristt).
ChimerMarker software also has a linked maternal cell contamination (MCC) application. ChimerMarker software is compatible with ABI®PRISM, Applied Biosystems® SeqStudio, and Promega Spectrum Compact CE Systems genetic analyzers, and custom primers or commercially available human identification chemistries for STR genotyping (including Identifiler®, GlobalFiler™ 6-Dye™, PowerPlex®16, PowerPlex®ESI, GenePrint® 24, PowerPlex®Fusion 5 and 6 Dye, Investigator®24Plex). Chimerism analysis is completely integrated to the main analysis screen, removing the error-prone step of data transfer from genotyping software to calculation spread sheets.
Using ChimerMarker gives 85% faster analysis! (paper)
Click here to read “Analytic Approach for Complete and Mixed Engraftment Analysis using Commercial Software”, presented at Applied Molecular Pathology meetings 2016 by Lea F Surrey MD, Jennifer R Smith BA, Daniel Lubin MD, Vivianna M. Van Deerlin MD PhD, and Christopher D Watt MD PhD.
ChimerMarker Webinars
- ChimerMarker Introduction (view on
 )
) - Calibrating panels in ChimerMarker (view on
 )
) - ChimerMarker Concordance with Best Practice Guidlines (view on
 )
) - Longitudinal Monitoring Using ChimerMarker Software (view on
 )
) - Maternal Cell Contamination (view on
 )
) - Double Donor (view on
 )
)
Application Notes:
- ChimerMarker® Software for Automated Analysis, Chimerism Detection, Quantification
and Monitoring from Short Tandem Repeat (STR) DNA in Post-Transplant Samples App Note (PDF) - Automated Chimerism Analysis Using ChimerMarker® Software: A Concordance Study (PDF)
> View the latest citations of ChimerMarker Software
Workflow
*Time study reported by Franciscan Alliance, St. Francis Health, Indianapolis, IN
Complete Chimerism Detection, Quantification and Analysis in one program
Software Applications
- Compliance with Best Practice Guidelines
- Validation and Implementation Study
(Penn Medicine/Children's Hospital of Philadelphia) - Accurate genotyping and informative locus flagging
- Single Donor Cases
- Double Donor Cases
- Longitudinal Monitoring
- Maternal Cell Contamination (MCC)
Time-Saving Linked Tools
Technical Support
The supporting documents available for this product can be downloaded below.