dam–/dcm– Competent E. coli
Product information| Code | Name | Size | Quantity | Price | |
|---|---|---|---|---|---|
C2925I |
dam-/dcm- Competent E. coli |
6 x 0.2 ml | - | Unavailable in your region | |
C2925H |
dam-/dcm- Competent E. coli |
20 x 0,05 ml | - | Unavailable in your region |
dam–/dcm– Competent E. coli
Product Introduction
Methyltransferase deficient E. coli cells suitable for growth of plasmids free of Dam and Dcm methylation
- No dry ice surcharge on competent cell shipments
- Outgrowth medium included
- Free of animal products
| Catalog # | Size | Concentration |
|---|---|---|
| C2925H | 20 x 0.05 ml | |
| C2925I | 6 x 0.2 ml |
- Product Information
- Protocols, Manuals & Usage
- Tools & Resources
- FAQs & Troubleshooting
- Citations & Technical Literature
- Quality, Safety & Legal
- Other Products You May Be Interested In
Product Information
Description
Highlights
- Grow plasmids free of Dam and Dcm methylation
- Free of animal products
- Phage T1 resistant (fhuA31)
- Activity of nonspecific endonuclease I (endA1) eliminated for highest quality plasmid preparations
- K12 Strain
Transformation Efficiency
1-3 x 106 cfu/μg pUC19 DNA Methyltransferase deficient chemically competent E. coli cells suitable for growth of plasmids free of Dam and Dcm methylation. Note that dam- strains are not recommended as a host for primary cloning/ligation. The dam mutation can result in an increased mutation rate in the cell and a reduction in the transformation efficiency. DNA should be maintained in a dam+ strain unless there is a specific need for DNA free of Dam or Dcm methylation.Genotype
ara-14 leuB6 fhuA31 lacY1 tsx78 glnV44 galK2 galT22 mcrA dcm-6 hisG4 rfbD1 R(zgb210::Tn10) TetS endA1 rspL136 (StrR) dam13::Tn9 (CamR) xylA-5 mtl-1 thi-1 mcrB1 hsdR2- This product is related to the following categories:
- Cloning Competent Cell Strains
- This product can be used in the following applications:
- High-throughput cloning and automation solutions,
- Transformation
Reagents Supplied
Reagents Supplied
The following reagents are supplied with this product:
| NEB # | Component Name | Component # | Stored at (°C) | Amount | Concentration | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||
Properties & Usage
Antibiotic for Plasmid Selection
| Antibiotics for Plasmid Selection | Working Concentration |
|---|---|
| Ampicillin | 100 µg/ml |
| Carbenicillin | 100 µg/ml |
| Kanamycin | 30 µg/ml |
| Tetracycline | 15 µg/ml |
Shipping Notes
- Ships on dry ice
Antibiotic Resistance
- str
- nit
- cam
Features
- Dam/Dcm methyltransferase free plasmid growth
Application Features
Related Products
Companion Products
- NEB® 10-beta Competent E. coli (High Efficiency)
- NEB® 5-alpha Competent E. coli (Subcloning Efficiency)
- NEB® 5-alpha F' Iq Competent E. coli (High Efficiency)
- NEB® Turbo Competent E. coli (High Efficiency)
- NEB® 5-alpha Competent E. coli (High Efficiency)
Materials Sold Separately
Product Notes
- CAUTION: This product contains DMSO, a hazardous material. Review the MSDS before handling.
- STORAGE AND HANDLING: Competent cells should be stored at -80°C. Storage at -20°C will result in a significant decrease in transformation efficiency. Cells lose efficiency whenever they are warmed above -80°C, even if they do not thaw.
Protocols, Manuals & Usage
Protocols
Usage & Guidelines
Application Notes
Tools & Resources
Selection Charts
Web Tools
FAQs & Troubleshooting
FAQs
- Can I transform unmethylated DNA into dam-/dcm- competent E.coli (C2925)?
- Does New England Biolabs offer a methyltransferase free strain of Competent E.coli?
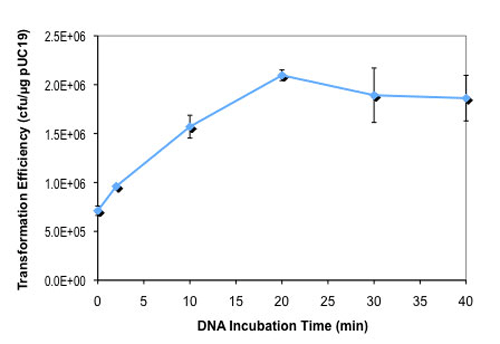
- How long should I incubate cells on ice after DNA has been added (NEB #C2925H and NEB #C2925I)?
- Is dam-/dcm- (NEB #C2925H/C2925I) resistant to kanamycin?
- Is the DNA yield lower using dam-/dcm- strain (C2925)?
- What are the solutions/recipes (C2925)?
- What are the strain properties (C2925)?
- What is the difference between NEB #C2925H and NEB #C2925I?
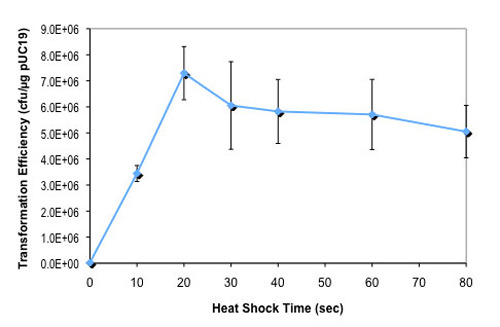
- What is the optimal heat shock time for this strain (NEB #C2925H and NEB #C2925I)?
- Which strain of Competent E.coli should I use for general cloning?
- Why were the colonies at different size after transformation of dam-/dcm- competent E.coli (C2925)?
- How should I calculate the transformation efficiency (C2925)?
- Can I store competent cells at -20°C instead of -80°C?
- Which kind of transformation tubes should be used?
- What volume of DNA can be added into competent cells?
- What is the shelf life for this strain (NEB #C2925H and NEB #C2925I)?
- Are NEB's competent cells compatible with the “Mix & Go" protocol?
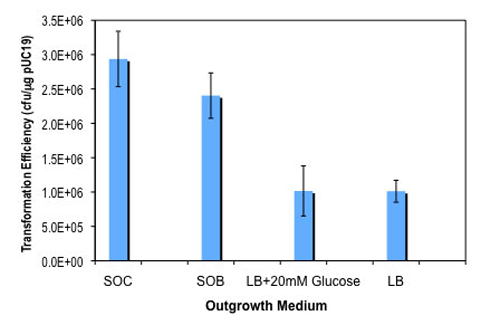
- How should I store SOC Outgrowth Medium? The SOC I received with my competent cells recommends storage at either room temperature or 4°C, however, when I purchase it as a stand alone product, it recommends storing it at 4°C. Which is better?
- How should fragments be prepared for assembly using NEBuilder HiFi?
Citations & Technical Literature
Citations
Additional Citations
Quality, Safety & Legal
Quality Assurance Statement
Quality Control tests are performed on each new lot of NEB product to meet the specifications designated for it. Specifications and individual lot data from the tests that are performed for this particular product can be found and downloaded on the Product Specification Sheet, Certificate of Analysis, data card or product manual. Further information regarding NEB product quality can be found here.Specifications
The Specification sheet is a document that includes the storage temperature, shelf life and the specifications designated for the product. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]Certificate Of Analysis
The Certificate of Analysis (COA) is a signed document that includes the storage temperature, expiration date and quality controls for an individual lot. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]_[Lot Number]- C2925H_I_v1_0561607
- C2925H_I_v1_0501502
- C2925H_I_v1_0561608
- C2925H_I_v1_0571608
- C2925H_I_v1_0521508
- C2925H_I_v1_0571610
- C2925H_I_v1_0551609
- C2925H_I_v1_0561610
- C2925H_I_v1_0541611
- C2925H_I_v1_0551611
- C2925H_I_v1_0581611
- C2925H_I_v1_0591611
- C2925H_I_v1_0541701
- C2925H_I_v1_0551701
- C2925H_I_v1_0541703
- C2925H_I_v1_0591703
- C2925H_I_v1_0591704
- C2925H_I_v1_0541705
- C2925H_I_v1_0581706
- C2925H_I_v1_0541603
- C2925H_I_v1_0561604
- C2925H_I_v1_0581707
- C2925H_I_v1_0581708
- C2925H_I_v1_0581710
- C2925H_I_v1_0591707
- C2925H_I_v1_0591708
- C2925H_I_v1_0611706
- C2925H_I_v1_0611709
- C2925H_I_v1_0611710
- C2925H_I_v1_0611712
- C2925H_I_v1_0581712
- C2925H_I_v1_0611801
- C2925H_I_v1_0601801
- C2925H_I_v1_0611803
- C2925H_I_v1_0601803
- C2925H_I_v1_0621803
- C2925I_v1_10011528
- C2925H_v1_10012789
- C2925I_v1_10013424
- C2925I_v1_10015495
- C2925H_v1_10015846
- C2925I_v1_10018135
- C2925H_v1_10020246
- C2925I_v1_10021088
- C2925I_v1_10024318
- C2925H_v1_10024963
- C2925H_v1_10026473
- C2925I_v1_10027603
- C2925H_v1_10029671
- C2925I_v1_10030908
- C2925H_v1_10033407
- C2925I_v1_10033920
- C2925H_v1_10037025
- C2925I_v1_10036364
- C2925H_v1_10041319
- C2925I_v1_10039962
- C2925H_v1_10042684
- C2925I_v1_10043190
- C2925H_v1_10045315
- C2925H_v1_10047054
- C2925I_v1_10046412
- C2925H_v1_10049344
- C2925I_v1_10049970
- C2925H_v1_10052234
- C2925I_v1_10052474
- C2925I_v1_10055256
- C2925H_v1_10056411
- C2925H_v1_10059033
- C2925I_v1_10058462
- C2925I_v1_10061589
- C2925H_v1_10063266
- C2925H_v1_10066361
- C2925I_v1_10066637
- C2925H_v1_10069842
- C2925I_v1_10069835
- C2925H_v1_10076058
- C2925I_v1_10076322
- C2925I_v1_10077686
- C2925H_v1_10079070
- C2925I_v1_10079069
- C2925I_v1_10081195
- C2925I_v1_10082918
- C2925H_v1_10083735
- C2925I_v1_10087560
- C2925H_v1_10088591
- C2925I_v1_10093045
- C2925H_v1_10093202
- C2925H_v1_10100036
- C2925I_v1_10100037
- C2925H_v1_10103121
- C2925I_v1_10103369
- C2925H_v1_10108149
- C2925I_v1_10108890
- C2925H_v1_10111109
- C2925H_v1_10115076
- C2925I_v1_10115136
- C2925H_v1_10118474
- C2925I_v1_10120367
- C2925H_v1_10122543
- C2925I_v1_10125857
- C2925H_v1_10129883
- C2925I_v1_10134347
- C2925H_v1_10138508
- C2925H_v1_10141120
- C2925I_v1_10144143
- C2925H_v1_10147010
- C2925I_v1_10150252
- C2925H_v1_10153042
- C2925H_v1_10158428
- C2925I_v1_10159145
- C2925H_v1_10164330
- C2925I_v1_10164776
- C2925H_v1_10166787
- C2925H_v1_10170681
- C2925I_v1_10173939
- C2925H_v1_10174676
- C2925H_v1_10177685
- C2925I_v1_10179281
- C2925H_v1_10181098
- C2925H_v1_10183461
- C2925I_v1_10183462
- C2925H_v1_10187594
- C2925I_v1_10188356
- C2925H_v1_10193330
- C2925I_v1_10196214
- C2925H_v1_10198494
- C2925H_v1_10203040
- C2925I_v1_10203627
- C2925H_v1_10207474
- C2925I_v1_10209797
- C2925H_v1_10212260
- C2925I_v1_10215680
- C2925H_v1_10225943
- C2925I_v1_10225537
- C2925I_v1_10230141
- C2925H_v1_10230788
- C2925H_v1_10236195
- C2925I_v1_10237224
- C2925H_v1_10240207
- C2925I_v1_10244782
- C2925H_v1_10245647
- C2925H_v1_10250179
- C2925I_v1_10250758
- C2925H_v1_10254818
- C2925I_v1_10254821
- C2925H_v1_10261005
- C2925I_v1_10264012
- C2925H_v1_10267487
- C2925I_v1_10274078
- C2925H_v1_10276522
- C2925H_v1_10281535
- C2925I_v1_10284575
- C2925H_v1_10290871
- C2925I_v1_10293833
- C2925H_v1_10298960
- C2925H_v1_10305990
- C2925I_v1_10306941
Safety DataSheets
The following is a list of Safety Data Sheet (SDS) that apply to this product to help you use it safely.dam–/dcm– Competent E. coli
pUC19 Vector
SOC Outgrowth Medium
Legal and Disclaimers
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email [email protected].This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.
New England Biolabs (NEB) is committed to practicing ethical science – we believe it is our job as researchers to ask the important questions that when answered will help preserve our quality of life and the world that we live in. However, this research should always be done in safe and ethical manner. Learn more.
The supporting documents available for this product can be downloaded below.
Protocol
Transformation Protocol (C2925)
Protocol
Transformation Protocol (C2925)
Protocol
Transformation Protocol (C2925)
Protocol
Transformation Protocol (C2925)




