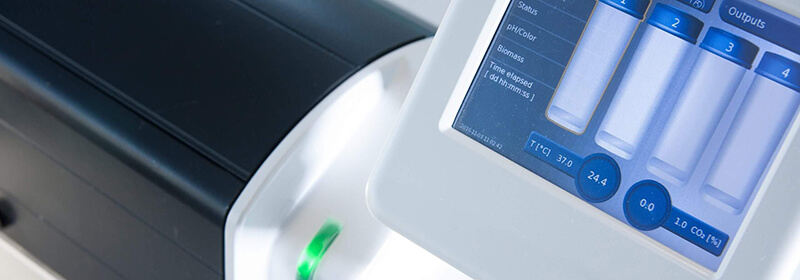
The CERO 3D Incubator & Bioreactor is a revolutionary instrument creating optimal cell culture environment. It offers a special 3D cell culture technology that monitors and controls temperature, pH and carbon dioxide levels. This is ideal for stem cells, spheroids, organoids and even tissues.
The instrument improves viability and maturation with minimized apoptosis and necrosis. The CERO has no shear forces, no requirement for embedding substrate, thus providing maximum homogeneity and allowing long-term cultivation for more than one year. This will significantly reduce your running costs.
 The individually controlled CEROtubes provide highest biomass yields. With volumes up to 50 ml, the CEROtubes have small fins and a flat bottoms allowing mild cultivation conditions in a standardized and reproducible way, with minimum handling requirements.
The individually controlled CEROtubes provide highest biomass yields. With volumes up to 50 ml, the CEROtubes have small fins and a flat bottoms allowing mild cultivation conditions in a standardized and reproducible way, with minimum handling requirements.
The CERO allows for simplified scale-up, automation platforms, and cost reductions. This is due to its distinct advantages of easy to set-up and simple workflows and minimum hands-on time that can be down to less than 2 minutes per day.
Applications
The system can be used for a wide variety of applications, from stem cell expansion projects in biobanks, cell-based drug discovery, toxicity testing and regenerative medicine. For stem cells, the CERO is able to differentiate in 3 germ layers, providing homogeneous iPSC and ESC 3D aggregates. Your cells can be easily processed directly for differentiation such as for organoids or spheroids as a downstream application in 3D or 2D assay development and avoid technical limitations of long-term culture with high efficiency and standardization.
Some example applications
Beating Cardiac Bodies – a complete workflow
Stem cell derived cardiomyocytes gain more and more attention in the field of cardiovascular research, disease modeling and drug development.
The CERO 3D Incubator & Bioreactor platform allows the workflow with stem cell expansion in homogeneous aggregates followed by direct induction into a high number of beating cardiac bodies. The expansion of pluripotent stem cells and subsequent cardiac induction/differentiation in CERO 3D results in a much higher cell quality, homogeneity, integrity and yield compared to traditional orbital shakers.
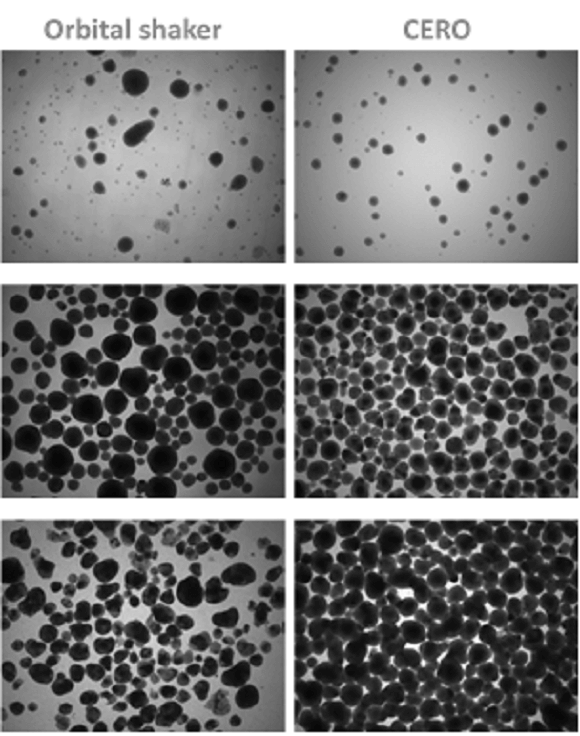
CERO 3D versus Orbital shaker – differentiation of murine embryonic stem cell derived cardiomyocytes 3, 8 and 13 days after cardiac induction.
- Homogeneous 3D aggregates in suspension
-
Expansion and differentiation in the same CEROtube
-
Easy to handle & standardize
-
Highest yield
-
No substrate required
-
More relevance for drug treatments
-
More relevance for disease modeling
Purified and homogeneous cardiac bodies generated in CERO 3D Incubator & Bioreactor are a perfect tool for disease modeling and drug discovery. The pure cardiomyocytes can also be used in 2D downstream monolayer applications.
From Adult Stem Cells to Organoids
The CERO 3D Incubator & Bioreactor is a revolutionary technology for production of organoids from pluripotent or adult stem cells. It offers an efficient, standardizable way to generate and maintain high yields of homogeneous organoids used as a tool in cancer research. Remarkably, the organoids have also a predictive capacity for in vivo response.
- Reduced costs and time
-
Increase size & lifespan
-
Optimal nutrition & gas diffusion
-
Free floating, no shear forces
-
High homogeneity and yield
-
Multiplexing organoidogenesis
Increase cellular, structural and functional complexity of organoids.
|
|
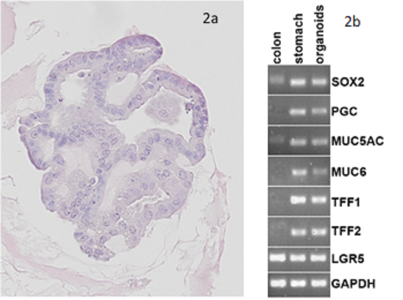
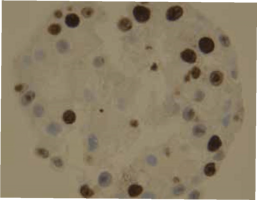
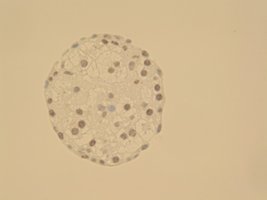
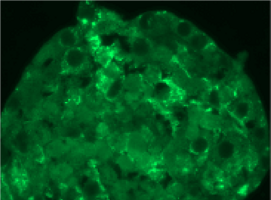
| HE staining of Gastric organoids (2a) showing single layer of epithelia cells composed by the different cell types found in the stomach as seen in the PCR results (2b): expression of gastric mucins MUC5AC and MUC6, trefoil factors and pepsinogen, for instance. There are also stem cells markers like Lgr5 and Sox2. |
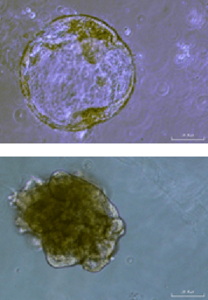
Gastric organoids (bright field) at day 7 and after splitting on day 22 expanding from small cysts that expand to bigger spheres |
Hepatitis Virus Research Model - Spheroids
The advent of long-term three dimensional cell culture holds a great promise in disease modeling and drug discovery. The cells kept in a 3D environment have the ability to mimic tissue-like structures more efficiently than in traditional 2D monolayer cultures. However, many scientist are struggling with many technical limitations when working with spheroids in long-term cultures.
The CERO 3D Incubator & Bioreactor is a revolutionary technology enabling scientists to perform experiments they were not able to do before.
- No necrosis and apoptosis
- Viability in long-term culture > 80 days
- Improved maturation
- Long-term proliferation
- High homogeneity
- High yield
- Spheroids from HepG2 cells (hepatocyte cell line) cultivated in CERO 3D for >80 days.
|
|
|
| Cells are positive for cell proliferation marker KI67 |
Cells are negative for apoptosis marker Casp.cl.3 |
Cells are positive for albumin |
More example applications can be found on the OLS website.
Highlights of the CERO
- Improved viability and maturation
- No embedding substrate required
- Significantly reduced apoptosis & necrosis
- No shear forces
- Long-term cultivation for > 1 year
- Significant reduced running costs
Meet the CERO 3D
Publications
“Stange, Katja, Amir Keric, Andreas Friese, and Monika Röntgen.
Preparation of Spheroids from Primary Pig Cells in a Mid-Scale Bioreactor Retaining Their Myogenic Potential
Cells 2022; 11, no. 9: 1453.”
DOI: 10.3390/cells11091453
“Saskia Altmaier, InaMeiser, Emilie Lemesre, Benjamin Chanrion, Rachel Steeg, Lidia Elena Leonte, Bjørn Holst, Boye Schnack Nielsen, Christian Clausen, Katharina Schmidt, Anne Marie Vinggaard, Heiko Zimmermann, Julia Christiane Neubauer, Mikkel AabechRasmussen
Human iPSC-derived hepatocytes in 2D and 3D suspension culture for cryopreservation and in vitro toxicity studies,
Reproductive Toxicology. Volume 111, August 2022, Pages 68-80”
DOI: 10.1016/j.reprotox.2022.05.005
“Chee Keong Kwok, Isabelle Sébastien, Krithika Hariharan, Ina Meiser, Jeanette Wihan, Saskia Altmaier, Isabell Karnatz, Alexander Feile, Alfredo Cabrera-Socorro, Mikkel Rasmussen, Bjørn Holst, Julia C. Neubauer, Christian Clausen, Catherine Verfaillie, Andreas Ebneth, Mattias Hansson, Rachel Steeg, Heiko Zimmermann
Scalable expansion of iPSC and their derivatives across multiple lineages
Reprod Toxicol. 2022 May 17;S0890-6238(22)00068-5.”
DOI: 10.1016/j.reprotox.2022.05.007
“Mathews, M., Wißfeld, J., Flitsch, L.J., Shahraz, A., Semkova, V., Breitkreuz, Y., Neumann, H., Brüstle, O. Reenacting neuroectodermal exposure of hematopoietic progenitors enables scalable production of cryopreservable iPSC-derived human microglia. Stem Cell Rev Rep. 2022.”
DOI: 10.1007/s12015-022-10433-w
More publications. Accessories CERO system
Learn more about the instruments from OMNI Life Sciences (OLS)