pCLIPf Vector
Product information| Code | Name | Size | Quantity | Price | |
|---|---|---|---|---|---|
N9215S |
pCLIPf Vector |
20 µg ( 0.5 mg/ml ) | - | Unavailable in your region |
pCLIPf Vector
Product Introduction
pCLIPf Vector is a mammalian expression plasmid intended for the cloning and stable or transient expression of CLIP-tag® protein fusions in mammalian cells. This plasmid encodes CLIPf, a CLIP-tag protein, which is expressed under control of the CMV promoter.
- View sequence details
- The expression vector has an IRES (internal ribosome entry site) and a neomycin resistance gene downstream of the CLIPf for the efficient selection of stable transfectants
- pCLIPf Vector contains two multiple cloning sites to allow cloning of the fusion partner as a fusion to the N- or C-terminus of the CLIPf
- pCLIPf contains an improved version of CLIP-tag
- CLIPf displays faster kinetics in in vitro labeling than CLIP-tag, and fast, specific and efficient labeling in live and fixed cell applications
| Catalog # | Size | Concentration |
|---|---|---|
| N9215S | 20.0 µg | 0.5 mg/ml |
- Product Information
- Protocols, Manuals & Usage
- Tools & Resources
- FAQs & Troubleshooting
- Citations & Technical Literature
- Quality, Safety & Legal
Product Information
Description
pCLIPf Vector is a mammalian expression plasmid intended for the cloning and stable or transient expression of CLIP-tag protein fusions in mammalian cells. This plasmid encodes CLIPf, a CLIP-tag protein, which is expressed under control of the CMV promoter. The expression vector has an IRES (internal ribosome entry site) and a neomycin resistance gene downstream of the CLIPf for the efficient selection of stable transfectants. pCLIPf Vector contains two multiple cloning sites to allow cloning of the fusion partner as a fusion to the N- or C-terminus of the CLIPf.The CLIP-tag is a novel tool for protein research, allowing the specific, covalent attachment of virtually any molecule to a protein of interest. The CLIP-tag is a small protein based on human O6-alkylguanine-DNA-alkyltransferase (hAGT). CLIP-tag substrates are derivatives of benzylcytosine (BC). In the labeling reaction, the substituted benzyl group of the substrate is covalently attached to the reactive cysteine of CLIP-tag forming a stable thioether link.
pCLIPf contains an improved version of CLIP-tag, termed CLIPf. CLIPf displays faster kinetics in in vitro labeling and fast, specific and efficient labeling in live and fixed cell applications, thereby rendering it a desired research tool for analysis of protein dynamics.
Although CLIP-tag is based on the same protein as SNAP-tag®, the benzylcytosine substrates form a separate class of substrates, different from the benzylguanine substrates recognized by SNAP-tag. CLIP-tag and SNAP-tag can be used for orthogonal simultaneous labeling.
There are two steps to using this system: sub cloning and expression of the protein of interest as a CLIPf fusion, and labeling of the fusion with the CLIP-tag substrate of choice. Cloning and expression of CLIPf fusion proteins are described in this document. The labeling of the fusion proteins with CLIP-tag substrates is described in the instructions supplied with the CLIP-tag substrates.

- This product is related to the following categories:
- Cellular Analysis Vectors Products,
- DNA Plasmids & Substrates Products,
- Protein Labeling Products,
- This product can be used in the following applications:
- CLIP Cell,
- CLIP Surface,
- Pulse Chase,
- In vivo Imaging,
- Receptor Internalization,
- Protein Localization, Protein Labeling Snap Clip
Reagents Supplied
Reagents Supplied
The following reagents are supplied with this product:
| NEB # | Component Name | Component # | Stored at (°C) | Amount | Concentration | |
|---|---|---|---|---|---|---|
Properties & Usage
Materials Required but not Supplied
- Tissue culture reagents and media
- Mammalian cell line(s)
- Transfection reagents
- CLIP-tag substrates
Lac Repressor on Plasmid
0Sequence Files
Fasta GenBankFeatures
BACKGROUND:
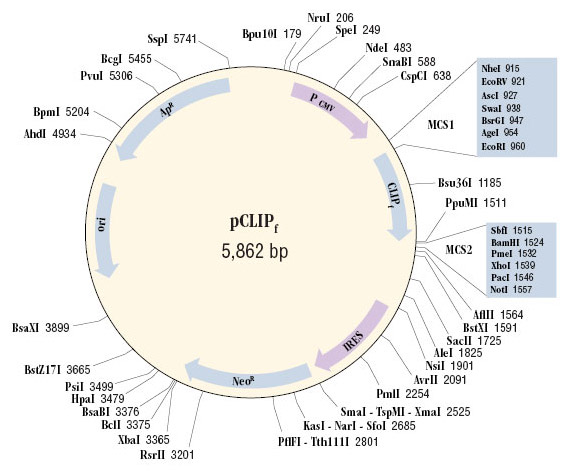
A plasmid map and the sequence of the cloning region can be found in this document. The complete plasmid sequence can be downloaded. This plasmid encodes the gene CLIPf which is a mutant form of the human gene for O6-alkylguanine-DNA-alkyltransferase (hAGT). The codon usage of the gene is optimized for expression in mammalian cells. In the plasmid sequence, the CLIPf gene is encoded from 969 bp to 1514 bp.
This plasmid is intended for the cloning and stable or transient expression of CLIP-tag protein fusions in mammalian cells. It is suitable for the efficient production of stable cell lines expressing CLIPf gene fusions. The plasmid contains the CMV promoter followed by the genes for CLIPf and neomycin resistance separated by the IRES of the encephalomyocarditis virus (ECMV), which permits the translation of two open reading frames from one messenger RNA; therefore after selection of stable mammalian cells for neomycin resistance, nearly all surviving colonies should stably express the CLIPf fusion protein. Unless the expression experiments require a pure population of cells, the pool of resistant cells can simply be used, otherwise cell clones can be isolated and characterized using standard procedures.
The plasmid contains the β-lactamase (Ampicillin resistance) gene for maintenance in bacteria. The gene of interest can be cloned upstream or downstream of the CLIPf coding sequence, as a fusion to the N- or C-terminus of the CLIP-tag. pCLIPf Vector can also be used as an expression control plasmid, expressing CLIPf alone, in which case the CLIP-tag protein is distributed throughout the cell. The CLIPf gene can be isolated from the plasmid using PCR or direct cloning in order to subclone it into a different vector of choice.
Product Notes
- Storage: pCLIPf Vector is supplied in TE buffer (10 mM Tris-HCl, pH 8.0, 1 mM EDTA) at a concentration of 0.5 µg/µl. Plasmid solutions can be stored at 4°C for up to one week. For long-term storage -20°C is recommended.
References
- Keppler, A. et al. (2003). Nat. Biotechnol. 21, 86.
- Gautier, A. et al. (2008). Chem. Biol. 15, 128.
- Keppler, A. et al. (2004). Proc. Natl. Acad. Sci. USA. 101, 9955.
- Maurel, D. et al. (2008). Nat. Methods. 5, 561.
- Jansen, L.E. et al. (2006). J. of Cell Biol. 176, 795.
- Krayl, M., Guiard, B. Paal, K. and Vous, W. (2006). Anal. Biol. Chem. 355, 81-89.
- Banala, S., Arnold, A. and Johnsson, K. (2008). ChemBio Chem. 9, 38-41.
Protocols, Manuals & Usage
Protocols
Application Notes
Tools & Resources
Selection Charts
Web Tools
FAQs & Troubleshooting
FAQs
Troubleshooting
Tech Tips
Citations & Technical Literature
Citations
Additional Citations
Quality, Safety & Legal
Quality Assurance Statement
Quality Control tests are performed on each new lot of NEB product to meet the specifications designated for it. Specifications and individual lot data from the tests that are performed for this particular product can be found and downloaded on the Product Specification Sheet, Certificate of Analysis, data card or product manual. Further information regarding NEB product quality can be found here.Specifications
The Specification sheet is a document that includes the storage temperature, shelf life and the specifications designated for the product. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]Certificate Of Analysis
The Certificate of Analysis (COA) is a signed document that includes the storage temperature, expiration date and quality controls for an individual lot. The following file naming structure is used to name these document files: [Product Number]_[Size]_[Version]_[Lot Number]Safety DataSheets
The following is a list of Safety Data Sheet (SDS) that apply to this product to help you use it safely.pCLIPf Vector
Legal and Disclaimers
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. The use of this product may require the buyer to obtain additional third-party intellectual property rights for certain applications. For more information, please email [email protected].This product is intended for research purposes only. This product is not intended to be used for therapeutic or diagnostic purposes in humans or animals.
New England Biolabs (NEB) is committed to practicing ethical science – we believe it is our job as researchers to ask the important questions that when answered will help preserve our quality of life and the world that we live in. However, this research should always be done in safe and ethical manner. Learn more.
Licenses
Cellular Imaging and Analysis (i.e., SNAP and CLIP products)
Notice to Buyer/User: The Buyer/User has a non-exclusive license to use this system or any component thereof for RESEARCH AND DEVELOPMENT ONLY. Commercial use of this system or any components thereof requires a license from New England Biolabs, Inc., 240 County Road, Ipswich, MA 01938. For detailed information see our Terms of Use .
These patents and patent applications are owned by Covalys, or owned by the Ecole Polytechnique Fédérale de Lausanne (EPFL) and exclusively licensed to Covalys and NEB.
The products and/or their use may be covered by one or more of the following patents and patent applications:
Labeling of Fusion Proteins with Synthetic Probes: US 8,227,602; US 8,623,627; and EP 2 049 499.
The supporting documents available for this product can be downloaded below.