
Polypropylene (PP) is the best plastic material for PCR tubes as it is chemically inert, resistant to solvents, and well suited for injection moulding - allowing for production of thin-walled tubes for optimum PCR results.
DNA has been shown to bind to PP tubes especially at high ionic strength, despite the very hydrophilic nature of this material. Different PP polymers are used for the production of PCR consumables and as they differ in their characteristics including surface charges, they consequently bind DNA in varying amounts.
DNA binding to PP surfaces has typically only been an issue for reaction tubes and storage vessels but not for PCR/qPCR tubes. DNA sticking to tube walls is either released during denaturation steps and/or remains accessible for amplification.
Nevertheless, due to a progressing volume miniaturisation and with new technologies such as Next Generation Sequencing (NGS), PCR/qPCR tubes are more and more also recommended for other applications which may require ultra-low DNA binding.
-
Low binding feature results from selected low-bind polymers, no coating is used to achieve the binding characteristics - No contamination or modification of the samples
-
Tested under a broad temperature range - Maximum DNA recovery after low temperature storage and high temperature incubation
-
No DNA loss during incubation steps and sample transfer in NGS sample prep and library construction - Ideally suited for sensitive applications with ultra-low DNA input
Experimental Data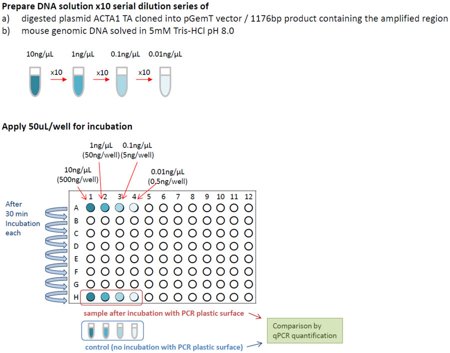
Fig 1, Binding of linear DNA to different PP polymers - Experimental set-up
Binding of linear DNA to different PP polymers
Experimental set-up
A tenfold DNA dilution series was created containing a linear 1176 bp product (digested from plasmid ACTA1, TA cloned into the pGemT vector) resulting in DNA concentrations of 10 ng/µl, 1 ng/µl, 0.1 ng/µl, and 0.010 ng/µl.
50 µl of each DNA concentration were applied to a PCR plate and incubated for 30 minutes at 37°C. The DNA was afterwards transferred to the next row of the PCR plate for additional 30 minute incubation. This procedure was repeated seven times so that the DNA was incubated in 8 different tubes for a total of 240 minutes. All transfer steps were performed with commercially available low-binding tips (Corning).
2 µl of each DNA concentration were subsequently subjected to qPCR analysis and compared to 2 µl of the original dilution series (total DNA input = 2 ng to 0.002 ng per qPCR run) using ACTA1 primer and probe set designed by IdT (Integrated DNA Technology).
Experimental results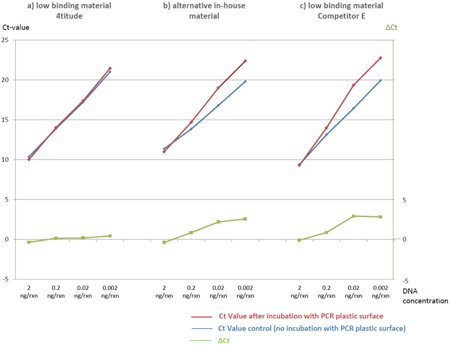
Fig 2, Binding of linear DNA to different PP polymers - qPCR comparison
Three different PCR plates were compared:
a) a PCR plate with tubes made of the 4titude low binding PP polymer
b) a PCR plate with tubes made of an alternative PP polymer and
c) a commercially available low-binding plate from competitor E.
Figure 2 shows Ct values obtained from the different DNA concentrations. The blue line shows the results for the control DNA not incubated in PCR tubes, the red line shows the results for DNA incubated in PCR tubes as described. The ΔCt is shown in green.
The red and the blue line should be identical (with ΔCt = 0) for plastic material which does not bind any detectable traces of DNA (minor differences may result from pipetting inaccuracies).
The 4titude low-binding material (a) shows almost an ideal pattern, the only small deviation from optimum results is a minor ΔCt of 0.44 cycles for the lowest DNA concentration of 0.002 ng/µl.
The alternative in-house PP polymer (b) tested in parallel revealed a significant DNA binding with a ΔCt of up to 2.57 cycles for the lowest DNA concentration. A surprisingly similar pattern with a ΔCt of up to 2.92 cycles was received from commercially available “low-binding” PCR plates from Competitor E (c).
The results clearly show the superior performance of the 4titude low binding PP polymer indicating the advantages for sensitive applications such as Next Generation Sequencing sample preparation.
Binding of genomic DNA to low binding PP polymer at different temperatures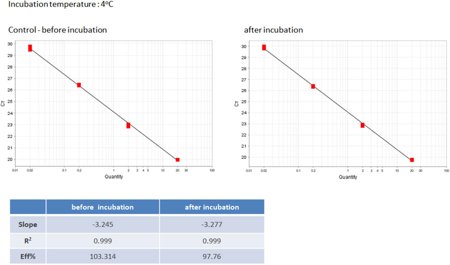
Fig 3, Binding of genomic DNA to low binding PP polymer at different temperatures - Exemplary standard curves
Further experiments were performed with the 4titude low binding PP polymer. The experiment described above was repeated with identical experimental conditions using mouse genomic DNA (dissolved in 5 mM Tris-HCl pH 8.0) instead of the ACTA1 fragment testing three different incubation temperatures (4°C as a typical storage temperature for reaction mixtures, 37°C as a typical temperature for enzyme reactions, and 65°C as a typical temperature for enzyme denaturation). qPCR comparisons were performed with identical DNA concentrations using KAPA SYBR Fast qPCR Master Mix.
Figure 3 shows an exemplary standard curve taken from the 4°C incubation showing the Ct value for 4 different input concentrations for incubated samples versus controls. The results for other temperatures were comparable.
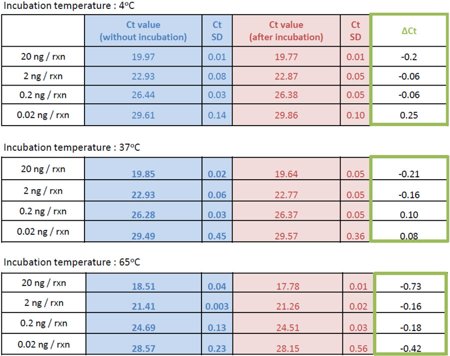
Table 1, Binding of genomic DNA to low binding PP polymer at different temperatures - Ct values for all different experimental conditions
Experimental data: the data comparing DNA adhesion at different temperatures was kindly provided by our Japanese cooperation partner Nippon Genetics.
Table 1 shows the Ct values for all different experimental conditions - confirming almost no differences between samples incubated in 8 different tubes for a total of 240 minutes and the control not subjected to PCR plastic surfaces.
This data set verifies the superior performance of the 4titude low binding PP polymer under different experimental conditions.
FrameStar® is covered by one or more of the following US patents or their foreign counterparts, owned by Eppendorf AG: US Patent Nos. 7,347, 977 and 6,340,589. FrameStar® is a registeredtrademark owned by 4titude®Ltd
The supporting documents available for this product can be downloaded below.



