Find your product
Advanced searchLogin
Webshop under construction
Due to technical maintenance the webshop is closed until January 3, 4. We wish you a successful 2025!
Welcome
Sign outPersonal information

Mission Bio
Unravel the threads of cancer’s complexity.
Company profile
Intricately woven, cancer and other illnesses challenge us with complexity. With Mission Bio, you can carefully tease apart each strand – genotype, phenotype, cell types, co-occurring mutations, zygosity, and structural changes – for a more holistic view of biology, revealing more meaningful sources of disease. Providing cancer researchers with the only single-cell multi-omics solution that simultaneously detects SNV, CNV, and protein data from the same cell.
Mission Bio is a company of passionate people dedicated to solving complex biological problems with precision engineering, innovative biochemistry, and supported bioinformatics.
In 2014, the company was spun out of Adam Abate’s lab for high-throughput biology at the University of California, San Francisco (UCSF). Mission Bio’s precision genomics platform is based on the pioneering droplet microfluidics work of Abate and his team. The mission is to help researchers and clinicians unlock single-cell biology to enable the discovery, development, and delivery of precision medicine.
Products
The first and only single-cell multi-omics platform developed to reveal biomarkers that help stratify patients, signal resistance, and predict relapse.
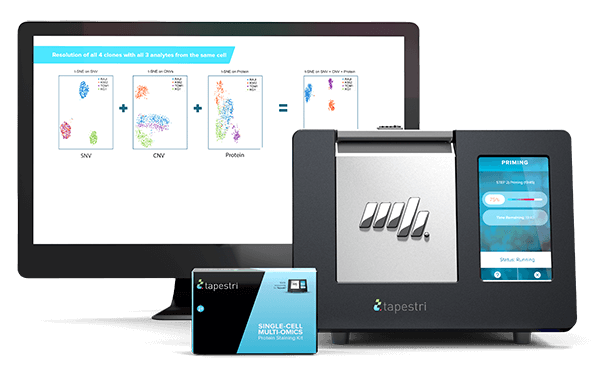
Tapestri Platform
An end-to-end workflow from sample preparation to visualization with publication-ready insights for single-cell analysis.
Learn more about the Tapestri Platform
Panels
Targeted panels provide focus on key regions of interest for efficient use of sequencing budget and analysis time.
Learn more about the Tapestri Panels
Learn more about Tapestri Oligos
Learn more about Tapestri Kits
Capabilities
- Single-cell Genomics (DNA)
-
Single-cell Multi-omics (DNA+)
- DNA + Protein - Gain a true multi-omics picture from genotype to phenotype.
Applications
-
Hematology.
Because single-cell insights are critical to determining the source of resistance, response, and relapse
The complexity of cancer is vast, and it is critical to measure the network of biological interactions accurately and minimize inferences. When DNA mutations can be analyzed with protein expression, you can discover clonal networks otherwise obfuscated by bulk methods done in parallel. By connecting genotype to phenotype at the single-cell level, you can gain comprehensive insights into disease progression, development of resistance, and minimal residual disease (MRD). Read more -
Solid Tumor.
Because clonal phylogeny and rare cell detection contribute to disease evolution
Solid tumors often show multiple levels of heterogeneity. This heterogeneity can contribute to molecular lesions play a role in driving cancer evolution and treatment response. With single-cell DNA sequencing, you can discern each patient's response profile to better study therapy response, resistance, and relapse. Read more -
Genome Editing.
Because zygosity and co-occurrence of multiple on and off-edit targets make a difference
Genome editing libraries are powerful tools to advance our understanding and treatment of complex diseases. With single-cell analysis, you can perform CRISPR validation and gain a holistic view of the sample with data integrated across multiple dimensions. Removing inferences, you can find answers to co-occurrence of on- and off-target edits, zygosity, transduction, and translocations with single-cell data. Characterize the workings of genome editing experiments in unprecedented detail. Read more -
Biomarker Development.
Because biomarker discovery helps stratify patients, signal resistance, and predict relapse
Understanding why some patients respond to therapies while others do not is critical to accelerating clinical trials and developing better treatments. Single-cell DNA and multi-omics analyses can untangle therapy resistance mechanisms that are impossible to obtain through traditional methodologies. The simultaneous detection of mutation profile and immunophenotype will help you discover new druggable targets, explore mechanisms of action, monitor patients, identify drug resistance as it develops, and stratify patients for clinical trials with greater efficiency. Read more